Depression is one of the most common and serious mental health conditions worldwide. It affects how people think, feel, and function in daily life. While traditional treatments such as antidepressants and psychotherapy help many individuals, they do not work for everyone. Some people experience limited improvement, delayed response, or unwanted side effects.
This is where clinical depression research trials become critically important.
Clinical trials are the foundation of medical progress. They help researchers evaluate new treatments, improve existing medications, and develop safer, more effective approaches to managing depression. For many individuals, participating in a depression clinical trial can offer access to innovative therapies while contributing to meaningful medical advancement.
In this comprehensive guide, you’ll learn:
- What clinical depression research trials are
- How they work
- Who qualifies
- Benefits and risks
- How they compare to traditional treatment
- The latest findings on depression trials
- Frequently asked questions
- How to participate
What Are Clinical Depression Research Trials?
Clinical depression research trials are carefully controlled medical studies designed to evaluate new treatments for depressive disorders. These studies may test:
- Investigational medications
- New uses for existing medications
- Combination therapies
- Novel mechanisms targeting brain chemistry
- Alternative delivery methods
- Advanced treatment strategies
Every FDA-approved antidepressant available today was once tested in clinical trials. Research studies are essential for ensuring treatments are safe, effective, and appropriate for patients.
Clinical depression studies are conducted under strict regulatory oversight, including:
- Institutional Review Boards (IRBs)
- FDA regulations
- Ethical standards for patient safety
- Informed consent requirements
Participation is voluntary, and patient safety is always the top priority.

Why Depression Research Is So Important
Depression affects millions of adults each year and remains a leading cause of disability. Despite available medications, many individuals:
- Do not respond to first-line antidepressants
- Experience treatment-resistant depression
- Struggle with side effects
- Need faster symptom relief
- Have co-occurring anxiety or other conditions
Clinical depression research trials help address these challenges by:
- Testing treatments with new biological targets
- Improving speed of symptom improvement
- Reducing unwanted side effects
- Developing personalized treatment approaches
- Exploring innovative therapeutic pathways
Without clinical trials, depression treatment would not evolve.
Types of Depression Studied in Clinical Trials
Depression is not a single condition. Research studies may focus on:
- Major Depressive Disorder (MDD)
- Treatment-Resistant Depression (TRD)
- Postpartum Depression
- Persistent Depressive Disorder
- Depression with anxiety features
- Bipolar depression (in certain protocols)
Each study has specific eligibility criteria based on diagnosis, symptom severity, and treatment history.
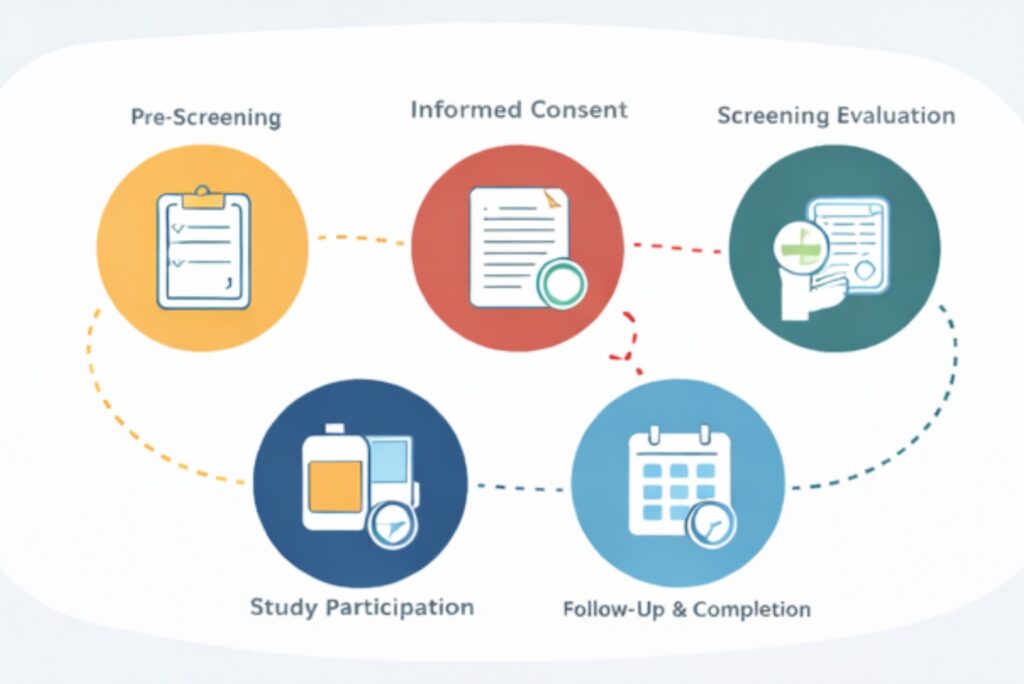
How Clinical Depression Research Trials Work
Understanding the process can reduce uncertainty and build confidence.
1. Pre-Screening
This typically involves:
- A brief phone or online questionnaire
- Review of basic eligibility criteria
- Discussion of medical history
If you appear eligible, you move to formal screening.
2. Informed Consent
Before participating, you receive detailed information about:
- Study purpose
- Procedures
- Potential risks and benefits
- Length of study
- Compensation (if applicable)
You may ask questions and take time to decide.
3. Screening Evaluation
This may include:
- Medical history review
- Depression assessments
- Physical exam
- Lab work
- Medication review
Not everyone who completes screening qualifies — eligibility is based on strict criteria to ensure safety and accurate results.
4. Study Participation
Participants may receive:
- Investigational medication
- Study-related care at no cost
- Placebo (in some trials)
- Regular mental health evaluations
- Close monitoring by experienced professionals
5. Follow-Up & Completion
After the study ends, participants may:
- Transition back to regular care
- Be referred for continued treatment
- Receive study completion compensation (if applicable)
Clinical Depression Trials vs Traditional Treatments
Many people wonder how clinical trials compare to standard care.
| Traditional Treatment | Clinical Depression Trial |
|---|---|
| FDA-approved medications | Investigational or next-generation therapies |
| Standard office visits | Structured, closely monitored visits |
| Insurance-based billing | Study-related care typically at no cost |
| Limited follow-up | Frequent mental health evaluations |
Clinical trials do not replace traditional care. Instead, they provide additional options — especially when standard treatments have not been effective.
Benefits of Participating in Depression Clinical Trials
Access to Innovative Treatments
Participants may receive therapies not yet available to the general public.
No-Cost Study Care
Study-related procedures and medication are typically covered.
Compensation
Some trials provide compensation for time and travel.
Expert Monitoring
Participants receive close supervision from medical professionals experienced in mental health research.
Contributing to Medical Advancement
By participating, you help improve treatment options for future generations.

Potential Risks of Clinical Depression Research Trials
Transparency is essential.
Potential risks may include:
- Side effects from investigational medication
- Lack of symptom improvement
- Time commitment
- Possibility of receiving placebo
However:
- Studies are monitored closely
- Safety reviews occur regularly
- You may withdraw at any time
Patient well-being is always prioritized.
Latest Findings on Depression Trials
Recent advancements in depression research include:
Faster-Acting Treatments
Some investigational therapies aim to reduce depressive symptoms more rapidly than traditional antidepressants.
Novel Mechanisms
New treatments target different brain pathways beyond serotonin.
Precision Medicine
Research is exploring how genetics and biology influence treatment response.
Expanded Treatment-Resistant Options
Innovative therapies are being studied for individuals who have not responded to multiple medications.
These latest findings on depression trials offer hope for more personalized and effective care in the future.

Who Can Participate in Depression Clinical Studies?
Eligibility depends on:
- Age requirements
- Confirmed depression diagnosis
- Current symptom severity
- Medication history
- Overall health status
Some studies require participants to:
- Currently take antidepressants
- Have failed prior treatments
- Be medication-free
A confidential pre-screening is the best way to determine eligibility.
Common FAQs About Clinical Depression Research Trials
1. Are clinical depression research trials safe?
Yes. Studies follow strict safety protocols, are reviewed by IRBs, and comply with FDA regulations.
2. Will I receive a placebo?
Some studies include placebo groups. This is explained clearly before enrollment.
3. Can I leave a study if I change my mind?
Yes. Participation is voluntary, and you may withdraw at any time.
4. Do I need insurance to participate?
Most study-related procedures are covered by the trial sponsor.
5. How long do depression trials last?
They may range from several weeks to several months, depending on the protocol.
6. Will my information remain confidential?
Yes. Participant privacy is protected under strict federal guidelines.
7. What if the treatment works for me?
Some studies may offer extension phases. After completion, your care team will discuss next steps.
8. Are depression trials only for severe cases?
No. Studies may enroll individuals with varying symptom severity levels.
9. Can I participate if I’m currently on medication?
Some trials allow stable medication use; others require changes. Eligibility depends on the protocol.
10. How do I start the process?
Contact the research site for a confidential pre-screening evaluation.
How Clinical Trials Advance Depression Treatment Options
Every advancement in depression care began with research participants.
Clinical trials:
- Identify safer medication profiles
- Improve response rates
- Shorten time to symptom relief
- Reduce relapse rates
- Expand options for treatment-resistant depression
Your participation directly contributes to the future of mental health treatment.
Is Participating in a Depression Clinical Trial Right for You?
You may consider exploring participation if:
- You continue to experience depressive symptoms
- Current medications are not providing relief
- You are interested in innovative treatments
- You want to contribute to medical progress
Speaking with a research professional can help you make an informed decision.
Take the Next Step with AIM Trials
At AIM Trials, our team is dedicated to:
- Providing compassionate care
- Offering clear information
- Ensuring patient safety
- Guiding you through every step
If you’re interested in learning more about clinical depression research trials, we invite you to:
- Complete a confidential pre-screening
- Speak with a study coordinator
- Explore currently enrolling studies
Your participation could help shape the future of depression treatment.